Anti-Inflammatory Activity of Root Ethanolic Extract of Harpagophytum procumbens on Human Peripheral Blood Mononuclear Cells and Screening of its Bioactive Compounds by GC-MS Method
Abstract
Background: A traditional African medicine consisting of an anti-inflammatory fame is the Devil s Claw, e.g. Harpagophytum procumbens. Its extracts have a broad application in Europe on the instances of musculoskeletal pain and low back pain as the therapy of such inflammatory issues. It has been found out that Harpagophytum procumbens is capable of alleviating pain and reducing the need of NSAIDs and possibly as analgesic and an anti-inflammatory agent, mainly in osteoarthritis.
Procedures: The powder was resieved with certified sieve. The volatility and polyphenols are the classes of phytochemicals whose biocompatibility and solubility dictated the choice of the solvents. An inert GCMS selective detector 6890N/5973 of Agilent Technologies, USA, was used to analyze by GC-MS. PBMCs isolation and harvesting of peripheral blood samples. The pretreatment and incubation of PBMCs with the three different doses of Harpagophytum procumbens was done after which the incubation time was permitted to elapse after two hours of which LPS (1.0 0g/mL) was added to stimulate the cells. The concentration of the pro-inflammatory mediators was measured after removing and storing at 70 25C the supernatants of the LPS-stimulated and the pretreatment PBMCs after 24 hours.
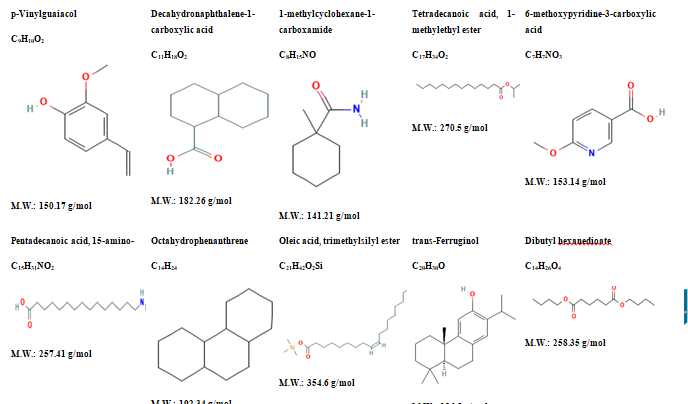
Results: GC-MS analysis belongs to the number of the common techniques that could be applied to study the chemical composition of the plant extracts, the one of H. procumbens included. The method is detectable to volatile and non-volatile root and plant extract constituents. Over 31 compounds are discovered as a result of the research. Speaking about the most frequent kind of chemicals we may point out the existence of p-Vinylguaiacol, Decahydronaphthalene, 1-methylcyclohexane-1-carboxamide, Tetradecanoic acid, 1-methylethyl ester, 6-methoxypyridine-3-carboxylic acid, Pentadecanoic acid, 15-amino, Octahydrophenanthrene, Oleic acid, trimethylsilyl ester, Cell viability of PBMCs after incubation with six concentrations (0, 10, 20, 40, 80, 160, and 320 ug/ml) of Harpagophytum procumbens at different time points (24, 48 and 72 h) was found to be 94.83+/-3.1, 91.13+/-3.7, 90.03+/-3.8, 87.03+/- The production of pro-inflammatory mediators (TNF-alpha, Interleukin-1b and Interleukin-6 Pg/mL respectively) was recorded as (3.96, 8.27, 4.91, 3.52, 30.46, 15.70, 12.50 and 9.58) Pg/mL respectively in the effect of Harpagophytum procumbens (0, 10, 60, 150, LPS But Interleukin-1 5.50, 10.49, 3.67, 1.50, 35.70, 16.88, 10.00 and 6.49 Pg/mL was recorded respectively. During the same time the Interleukin-6 levels were (35.00, 60.71, 53.98, 18.93, 180.01, 176.61, 144.00 and 133.58) Pg/mL respectively.
Full text article
References
Heinrich, M. et. al. (2012). Fundamentals of pharmacognosy and phytotherapy. Oxford: Elsevier Health Sciences, 336.
Wagner, H., Bladt, S. (2001). Plant drug analysis. Berlin: Springer, 384.
Chrubasik, S., Conradt, C., Roufogalis, B. D. (2004). Effectiveness ofHarpagophytum extracts and clinical efficacy. Phytotherapy Research, 18 (2), 187–189.
Brendler, T., Gruenwald, J., Ulbricht, C., Basch, E. (2006). Devil’s Claw (Harpagophytum procumbensDC). Journal of Herbal Pharmacotherapy, 6 (1), 89–126.
Lanhers, M.-C., Fleurentin, J., Mortier, F., Vinche, A., Younos, C. (1992). Anti-Inflammatory and Analgesic Effects of an Aqueous Extract ofHarpagophytum procumbens. Planta Medica, 58 (2), 117–123.
Andersen, M. L., Santos, E. H., Seabra, M. de L. V., da Silva, A. A., Tufik, S. (2004). Evaluation of acute and chronic treatments with Harpagophytum procumbens on Freund’s adjuvant-induced arthritis in rats. Journal of Ethnopharmacology, 91 (2-3), 325–330.
Nasiruddin, A. F., Akalanka, D., Singh, G. N., Easwari, T. S., Manoj, K. P. (2014). Analytical techniques in quality evaluation of herbal drugs. Asian Journal of Pharmaceutical research, 4 (3), 112–117.
Goldring, S.R.; Goldring, M.B. Changes in the osteochondral unit during osteoarthritis: Structure, function and cartilage bone crosstalk. Nat. Rev. Rheumatol.–644.
Turcotte, C.; Blanchet, M.R.; Laviolette, M.; Flamand, N. The CB2 receptor and its role as a regulator of inflammation. Cell. Mol. Life Sci. 2016, 73, 4449–4470.
Sophocleous, A.; Börjesson, A.E.; Salter, D.M.; Ralston, S.H. The type 2 cannabinoid receptor regulates susceptibility to osteoarthritis in mice. Osteoarthr. Cartil. 2015, 23, 1586–1594.
Chiou, L.C.; Hu, S.S.J.; Ho, Y.C. Targeting the cannabinoid system for pain relief? Acta Anaesthesiol. Taiwanica 2013, 51, 161–170.
Zoratti, C.; Kipmen-Korgun, D.; Osibow, K.; Malli, R.; Graier, W.F. Anandamide initiates Ca2+ signaling via CB2 receptor linked to phospholipase C in calf pulmonary endothelial cells. Br. J. Pharmacol. 2003, 140, 1351–1362.
Ferrara, A.L.; Piscitelli, F.; Petraroli, A.; Parente, R.; Galdiero, M.R.; Varricchi, G.; Marone, G.; Triggiani, M.; Di Marzo, V.; Loffredo, S. Altered metabolism of phospholipases, diacylglycerols, endocannabinoids, and N-Acylethanolamines in patients with mastocytosis. J. Immunol. Res. 2019.
Nagao, M.; Tanabe, N.; Manaka, S.; Naito, M.; Sekino, J.; Takayama, T.; Kawato, T.; Torigoe, G.; Kato, S.; Tsukune, N.; et al. LIPUS suppressed LPS-induced IL-1α through the inhibition of NF-κB nuclear translocation via AT1-PLCβ pathway in MC3T3-E1 cells. J. Cell. Physiol. 2017, 232, 3337–3346.
Dunn, S.L.; Wilkinson, J.M.; Crawford, A.; Bunning, R.A.D.; Le Maitre, C.L. Expression of Cannabinoid Receptors in Human Osteoarthritic Cartilage: Implications for Future Therapies. Cannabis Cannabinoid Res. 2016, 1, 3–15.
R. Medzhitov, Origin and physiological roles of inflammation, Nature 454 (7203) (2008) 428–435.
R. Medzhitov, Inflammation 2010: new adventures of an old flame, Cell 140 (6) (2010) 771–776.
L. Chen, et al., Inflammatory responses and inflammation-associated diseases in organs, Oncotarget 9 (6) (2018) 7204–7218.
A.H. Katsanos, et al., Biomarker Development in Chronic Inflammatory Diseases, Biomarkers for Endometriosis, 2017, pp. 41–75.
A. Shapouri-Moghaddam, et al., Macrophage plasticity, polarization, and function in health and disease, J. Cell. Physiol. 233 (9) (2018) 6425–6440.
B. Arosio, et al., Peripheral blood mononuclear cells as a laboratory to study dementia in the elderly, BioMed Res. Int. 2014 (2014) 169203.
A.S. Santos, et al., Prevalence of inflammatory pathways over Immuno-Tolerance in peripheral blood mononuclear cells of Recent-onset type 1 diabetes, Front. Immunol. 12 (2021) 765264.
C.-C. Liew, et al., The peripheral blood transcriptome dynamically reflects system wide biology: a potential diagnostic tool, J. Lab. Clin. Med. 147 (3) (2006) 126–132.
B. Reyn´es, et al., Peripheral blood cells, a transcriptomic tool in nutrigenomic and obesity studies: current state of the art, Compr. Rev. Food Sci. Food Saf. 17 (4) (2018) 1006–1020.
O. Pansarasa, et al., Biomarkers in human peripheral blood mononuclear cells: the state of the art in amyotrophic lateral sclerosis, Int. J. Mol. Sci. 23 (5) (2022).
M.A. Dobrovolskaia, S.N. Vogel, Toll receptors, CD14, and macrophage activation and deactivation by LPS, Microb. Infect. 4 (9) (2002) 903–914.
F. Naghibi, et al., Labiatae family in folk medicine in Iran: from ethnobotany to pharmacology, Iran. J. Pharm. Res. (IJPR) 4 (2) (2022) e128228.
S. Miraj, S. Kiani, Study of pharmacological effect of Mentha pulegium: a review, Der Pharm. Lett. 8 (9) (2016) 242–245.
Farshad H. Shirazi, Neda Ahmadi, Mohammad Kamalinejad, Evaluation of Northern Iran Mentha Pulegium L. Cytotoxicity, DARU Journal of Pharmaceutical Sciences, 2004, p. 5.
A.R. Abubakar, M. Haque, Preparation of medicinal plants: basic extraction and fractionation procedures for experimental purposes, J. Pharm. BioAllied Sci. 12 (1) (2020) 1–10.
V.R. Askari, et al., The influence of hydro-ethanolic extract of Portulaca oleracea L. on Th(1)/Th(2) balance in isolated human lymphocytes, J. Ethnopharmacol.194 (2016) 1112–1121.
Authors
Copyright (c) 2025 https://creativecommons.org/licenses/by/4.0/

This work is licensed under a Creative Commons Attribution 4.0 International License.