The Antibacterial, Antioxidant Properties and Natural Compound Screening Method of Cinnamomum zeylanicum Bark was Investigated through FTIR Testing
Abstract
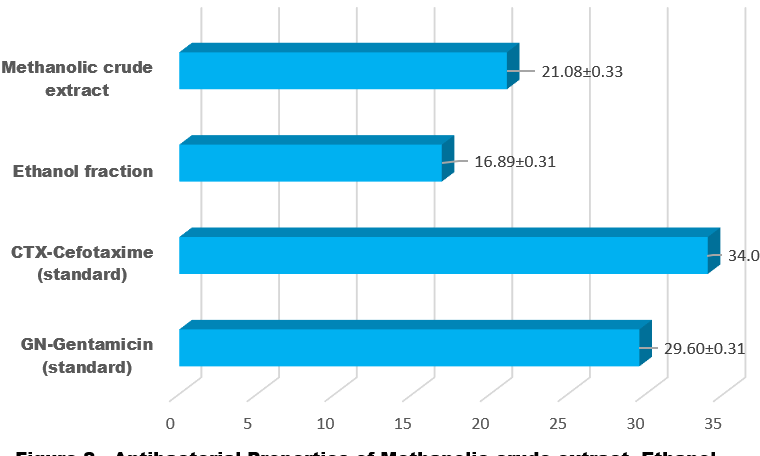
Both in vivo and in vitro studies have demonstrated that phytochemical components of photobiotic have potent antibacterial effects against Gram-positive and Gram-negative bacteria. There was a lack of data on cinnamon powder's minimum inhibitory concentration (MIC) and in vitro antibacterial effectiveness against different microorganisms. Thus, the purpose of this research was to compare the in vitro antibacterial activity and MIC of cinnamon powder extracts with that of conventional antibacterial medications against a range of microorganisms. The standards of CTX-Cefotaxime and GN-Gentamicin, as well as the methanolic crude extract and ethanol fraction of Cinnamomum zeylanicum bark extracts, have antibacterial activities that were measured at 21.08±0.33, 16.89±0.31, 34.00±0.32, and 29.60±0.31 respectively for Bacillus subtilis, and recorded 13.89±0.29, 17.00 ±0.32, 24.81±0.25, and 26.90±0.27 respectively for Enterococcus faecalis, while recorded 09.11±0.15, 19.75±0.32, 20.00±0.21 and 17.45±0.19 respectively for Streptococcus pyogenes, in the same time antibacterial activity recorded 15.00±0.30, 08.68±0.14, 27.90±0.29 and 26.04±0.29 respectively for Staphylococcus epidermidis and recorded 15.90±0.30, 14.06±0.29, 29.95±0.31 and 32.17±0.33 respectively for Staphylococcus aureus. FT-IR peak values of solid analysis of Cinnamomum zeylanicum Bark. Peak (Wave cm-ˡ, Intensity, Corr. Intensity, Base (H), Area, Bond, Type of Vibration, and Functional group assignment recorded 667.37, 70.282, 2.383, 667.01, 2.226 and Alkenes; 690.52, 71.720, 0.518, 713.66, 3.769, and Alkenes; 788.89, 77.439, 2.341, 821.68, 4.707, Alkenes; 999.13, 66.259, 1.402, 1006.84, 12.823, and Alkenes; 1029.99, 63.849, 0.209, 1031.92, 4.297, and alkyl halides; 1226.73, 82.894, 0.214, 1228.66, 2.659, and alkyl halides; 1417.68, 85.533, 0.939, 1427.32, 2.347, and alkyl halides; 1454.33, 85.135, 1.371, 1483.26, 2.052, and alkyl halides; 1516.05, 85.999, 0.843, 1521.84, 1.943, and Aromatic; 1539.20, 85.109, 1.482, 1571.99, 2.655, and Aromatic; 1645.28, 78.585, 0.511, 1647.21, 5.122, Amide; 2854.65, 85.991, 4.453, 2868.15, 2.326, and Alkane. The anti-oxidant properties of Cinnamomum zeylanicum were studied in methanol, ethanol extract, and standards, specifically looking at its ability to scavenge superoxide and nitric oxide radicals recorded 23.79±1.92, 19.84±1.75, and Quercetin (standard) 41.00±3.07 respectively of Superoxide radical scavenging. 43.90±3.08, 31.00±2.95, and Curcumin (standard) 65.00±4.97 respectively of Nitric oxide radical scavenging.
Full text article
References
De Castro ML, Garcıa-Ayuso LE. Soxhlet extraction of solid materials: an outdated technique with a promising innovative future. Anal. Chim. Acta. 1998;369(2):1-10.
Wayne PA. National committee for clinical laboratory standards. Performance standards for antimicrobial disc susceptibility testing.2002;12:1-53.
Wiegand I, Hilpert K, Hancock RE. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 2008;3(2):163-75
Sofia PK, Prasad R, Vijay VK, Srivastava AK. Evaluation of antibacterial activity of Indian spices against common foodborne pathogens. Int. J. Food Sci. Technol. 2007;42(8):910-15.
Gupta C, Garg AP, Uniyal RC, Kumari A. Comparative analysis of the antimicrobial activity of cinnamon oil and cinnamon extract on some food-borne microbes. Afr. J. Microbiol. Res. 2008;2(9):247-51.
Hoque MM, Inatsu M, Juneja V, Kawamoto S. Antimicrobial activity of cloves and cinnamon extracts against food borne pathogens and spoilage bacteria and inactivation of Listeria monocytogenes in ground chicken meat with their essential oils. Rep. Natʼl. Food Res. Inst. 2008;72 9-21.
Shafique M, Khan SJ, Naseen Z. Appraisal of nutritional status and antimicrobial activity of clove, kalvonji, cinnamon, black pepper and sweet basil. Pharmacologyonline. 2010;2:591-99.
Ababutain IM. Antimicrobial activity of ethanolic extracts from some medicinal plant. Aust. J. Basic Appl. Sci. 2011;5(11):678-83.
Mukhtar S, Ghori I. Antibacterial activity of aqueous and ethanolic extracts of garlic, cinnamon and turmeric against Escherichia coli ATCC 25922 and Bacillus subtilis DSM 3256. Int. J. Appl. Biol. Pharm. 2012;3(2):131-36.
Kapilan R. Determination of antibacterial activity of some important spices. Int. J. Res. Granthaalayah. 2015;3(10):57-60.
Upadhyaya S, Yadav D, Chandra R, Arora N. Evaluation of antibacterial and phytochemical properties of different spice extracts. Afr. J. Microbiol. Res. 2018;12(2):27-37.
Budhathoki S, Basnet A. Study of Antibacterial Activity of Household Spices. Int. J. Eng. Res. Appl. 2018;9(8):1700-1704.
Keloth S, Krishnamurthy K, Janardhanan J, Azeez S. Antimicrobial properties in bark and leaf extracts of four cinnamomum species. J. Evol. Med. Dent. Sci. 2018;7(5):683-89.
Abdulrasheed M, Ibrahim IH, Luka A, Maryam AA, Hafsat L, Ibrahim S, et al. Antibacterial effect of Cinnamon (Cinnamomum zeylanicum) bark extract on different bacterial isolates. J. Environ. Microbiol. Toxicol. 2019;7(1):16-20.
Islam MA, Islam MS, Hossain MA, Ahmed S, Aktaruzzaman M, Akanda MR, et al. In Vitro antibacterial activity of medicinal plants turmeric, cinnamon and clove against Gm (+Ve) and Gm (-Ve) bacteria. Int. J. Curr. Pharm. 2019;11(5):85-88.
Prajapati JA, Humbal BR, Sadariya KA, Bhavasar SK, Thaker AM. In vitro antibacterial sensitivity of cinnamon and clove oils against gram positive and gram-negative bacteria. J. Vet. Pharmacol. Toxicol. 2018;17(1):63-67.
Kavitha R, Valli C, Karunakaran R, Vijayarani K, Amutha R. Evaluation of antibacterial activity of some indian herbal extracts. Int. J. Curr. Microbiol. 2020;9(7):17-24.
Kris-Etherton PM, Hecker KD, Bonanome A, Coval SM, Binkoski AE, Hilpert KF, Etherton TD. Bioactive compounds in foods: their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002;113(9):71-88.
Manson MM. Cancer prevention–the potential for diet to modulate molecular signalling. Trends Mol. Med. 2003;9(1):11-18.
Surh YJ. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer. 2003;3(10):768-80
Burt S. Essential oils: their antibacterial properties and potential applications in foods-a review. Int. J. Food Microbiol. 2004;94(3):223- 53.
83. Qian, W.; Yang, M.; Wang, T.; Sun, Z.; Liu, M.; Zhang, J.; Zeng, Q.; Cai, C.; Li, Y. Antibacterial Mechanism of Vanillic Acid on Physiological, Morphological, and Biofilm Properties of Carbapenem-Resistant Enterobacter hormaechei. J. Food Prot. 2020, 83, 576–583.
84. Sethupathy, S.; Ananthi, S.; Selvaraj, A.; Shanmuganathan, B.; Vigneshwari, L.; Balamurugan, K.; Mahalingam, S.; Pandian, S.K. Vanillic acid from Actinidia deliciosa impedes virulence in Serratia marcescens by affecting S-layer, flagellin and fatty acid biosynthesis proteins. Sci. Rep. 2017, 7, 16328.
85. Yemis, G.P.; Pagotto, F.; Bach, S.; Delaquis, P. Effect of vanillin, ethyl vanillin, and vanillic acid on the growth and heat resistance of Cronobacter species. J. Food Prot. 2011, 74, 2062–2069.
14. Mamatova, A.S.; Korona-Glowniak, I.; Skalicka-Wozniak, K.; Jozefczyk, A.; Wojtanowski, K.K.; Baj, T.; Sakipova, Z.B.; Malm, A. Phytochemical composition of wormwood (Artemisia gmelinii) extracts in respect of their antimicrobial activity. BMC Complement. Altern. Med. 2019, 19, 288.
Bayoub K, Baibai T, Mountassif D, Retmane A, Soukri A. Antibacterial activities of the crude ethanol extracts of medicinal plants against Listeria monocytogenes and some other pathogenic strains. Afr. J. Biotechnol. 2010; 9(27):4251-58.
Ismail MM, Essam TM, Mohamed AF, Mourad FE. Screening for the antimicrobial activities of alcoholic and aqueous extracts of some common spices in Egypt. Int. J. Microbiol. Res. 2012;3(3):200-207.
Kamleshiya P, Meshram VG, Ansari AH. The in-vitro bacteriostatic potential of some traditionally used indian spices. Asian J. Res. Chem. 2012;5(4):492-96.
Liang Y, Li Y, Sun A, Liu X.Chemical compound identification and antibacterial activity evaluation of cinnamon extracts obtained by subcritical n‐butane and ethanol extraction. Food Sci. Nutr. 2019;7(6):2186-93.
Alagawany M, Farag MR, El-Kholy MS, El-Sayed SAA, Dhama K. Effect of resveratrol, cinnamaldehyde and their combinations on the antioxidant defense system and ATP release of rabbit erythrocytes: in vitro study. Asian J. Anim Vet. Adv. 2017;12:1-9.
Sinnhuber RO, Yu TC. The 2-thiobarbituric acid reaction, an objective measure of the oxidative deterioration occurring in fats and oils. J Japan Oil Chemist’s Soc 1977;26:259-67.
Li WQ, Wang J, Sun JF, Li WQ, Wang YJ, Zhang GJ. Shelflife prediction modeling of vacuum-packaged scallops on the kinetics of total volatile base nitrogen. Int J Food Sci Eng 2011;7:1-15.
Meng RG, Tian YC, Yang Y, Shi J. Evaluation of DPPH free radical scavenging activity of various extracts of Ligularia fischeri in vitro: a case study of Shaanxi region. Indian J Pharm Sci 2016;78:436-442.
Mathew S, Abraham TE. In vitro antioxidant activity and scavenging effects of Cinnamomum verum leaf extract assayed by different methodologies. Food Chem Toxicol 2006;44: 198-206.
Manchini-Filho J, Van- Koij A, Mancini DAP, Cozzolino FF, Torres RP. Antioxidant activity of cinnamon (Cinnamomum Zeylanicum, Breyne) extracts. Boll Chim Farm 1998;137: 443-7.
Truong DH, Nguyen DH, Ta NTA, Bul AV, Do TH, Nguyen HC. Evaluation of the use of different solvents for phytochemical constituents, antioxidants, and in vitro anti-inflammatory activities of Severinia buxifolia. J Food Qual 2019; 2019;8178294.
Cuong TV, Chin KB. Evaluation of Cudrania tricuspidata leaves on antioxidant activities and physicochemical properties of pork patties. Korean J Food Sci Anim Resour 2018;38: 889-900.
Singh N, Rajini PS, Free radical scavenging activity of an aqueous extract of potato peel. Food Chem 2004;85:611-6.
Widyawati PS, Tarsisius DWB, Fenny AK, Evelyn LW. Difference of solvent polarity to phytochemical content and antioxidant activity of Pluchea incicia less leaves extract. Int J Pharmacogn Phytochem Res 2014;6:850-5.
Georgieva L, Mihaylova D. Evaluation of the in vitro antioxidant potential of extracts obtained from Cinnamomum zeylanicum barks. Sci Work Russian Univer 2014;53:41-5.
Kamleshiya P, Meshram VG, Ansari AH. Comparative evaluation of antioxidant and free radical scavenging activity of aqueous and methanolic spice extracts. Int J Life Sci Pharm Res 2012;2:118-25.
Arisha SM, Sakr SA, Abd-Elhaseeb FR. Cinnamomum zeylanicum alleviate testicular damage induced by high fat diet in albino rats; histological and ultrastructural studies. Heliyon. 2020 Nov 1;6(11):1-14.
Castro JC, Pante GC, Centenaro BM, Almeida RTR De, Pilau EJ, Dias Filho BP, et al. Antifungal and antimycotoxigenic effects of Zingiber officinale, Cinnamomum zeylanicum and Cymbopogon martinii essential oils against Fusarium verticillioides. Food Addit Contam Part A. 2020;37(9):1531-41.
Ranjbar A, Ghasmeinezhad S, Zamani H, Malekirad AA, Baiaty A, Mohammadirad A, et al. Antioxidative stress potential of Cinnamomum zeylanicum in humans: a comparative cross-sectional clinical study. Therapy. 2006;3(1):113-7.
Ghosh T, Basu A, Adhikari D, Roy D, Pal AK. Antioxidant activity and structural features of Cinnamomum zeylanicum. 3 Biotech. 2015;5(6):939-47.
El-Baroty GS, Abd El-Baky HH, Farag RS, Saleh MA. Characterization of antioxidant and antimicrobial compounds of cinnamon and ginger essential oils. African J Biochem Res. 2010;4(6):167-74.
Dudonné S, Vitrac X, Coutiére P, Woillez M, Mérillon JM. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J Agric Food Chem [Internet]. 2009;57(5):1768-74.
Rao PV, Gan SH. Cinnamon: A multifaceted medicinal plant. Evidence-based Complement Altern Med. 2014;2014:1-13.
Singh R, Lawrence R, Lawrence K, Agarwal B, Gupta RK, Dar S. Antioxidant and Antibacterial Activity of Syzigium aromaticum, Zingiber officinale and Cinnamomum zeylanicum Essential Oils. Chem Sci Trans. 2015;4(1):239-45.
Borzoei A, Rafraf M, Niromanesh S, Farzadi L, Narimani F, Doostan F. Effects of cinnamon supplementation on antioxidant status and serum lipids in women with polycystic ovary syndrome. J Tradit Complement Med. 2018;8(1):128-33.
Wilson AA, Pierre MDJ, Leopold NT, Priya P, Nisha P. Stabilisation potentials of the essential oils of Thymus vulgaris L., Cinnamomum zeylanicum B. and Mentha piperita L. on palm olein at accelerated storage. African J Biotechnol. 2020;19(7):464-77.
Schmidt E, Jirovetz L, Buchbauer G, Eller GA, Stoilova I, Krastanov A, et al. Composition and antioxidant activities of the essential oil of cinnamon (Cinnamomum zeylanicum blume) leaves from Sri Lanka. J Essent Oil-Bearing Plants. 2006;9(2):170-82.
Cardoso-Ugarte GA, López-Malo A, Sosa-Morales ME. Cinnamon (Cinnamomum zeylanicum) essential oils. In: Preedy VR, editor. Essential Oils in Food Preservation, Flavor and Safety. Elsevier Inc.; 2016;4:339-47.